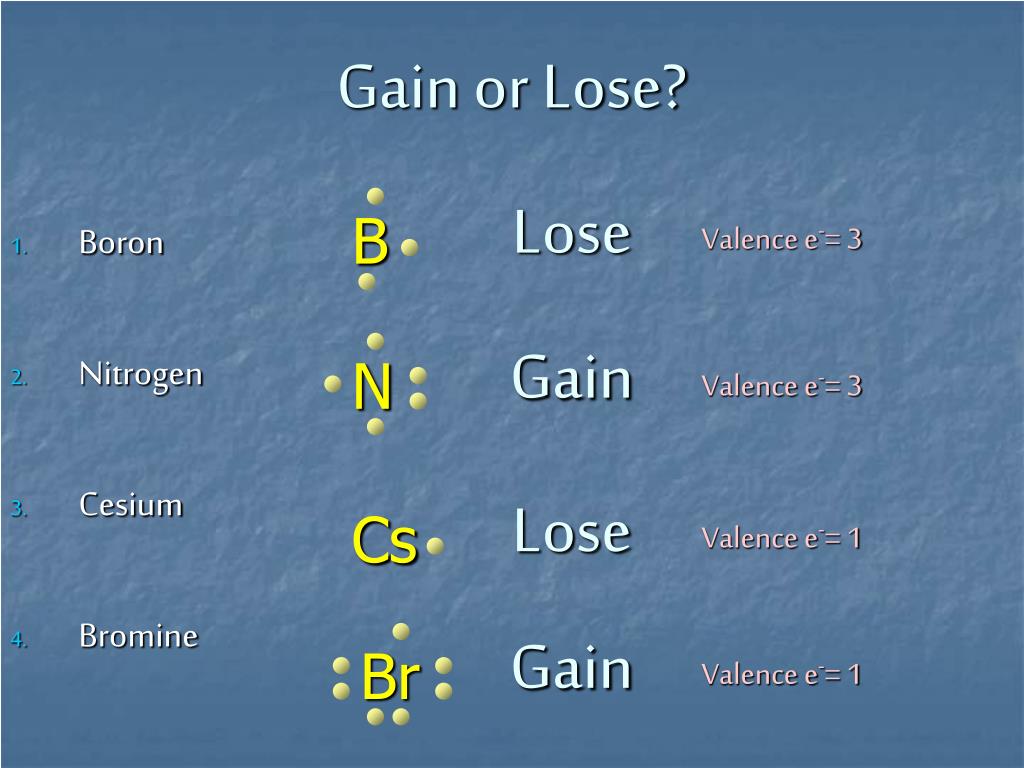
Chemistry
Which atom has the smallest valence p atomic orbital? -Carbon -Nitrogen -Sulfur Why? A. That atom has the highest electronegativity. B. That atom has the fewest number of electrons. C. That atom does not have electrons in pchemistry
elements below, indicate how each elements’ valence electrons act in a chemical reaction. Insert an L if an element loses electrons, a G if the element gains electrons, an E if the element can either lose or gain electrons, andAp Chem
explain each of the following observations using principles of atomic stucture and/or bonding. a] potassium has a lower first-ionization engergy than lithium. b] the ionic radius of N3- is larger than that of O2-. c] a calciumChemistry
Why would two atoms form a covalent bond instead of an ionic bond? Both atoms in a bond have similar electronegativity; thus, neither atom is willing to transfer their electrons. Instead, the atoms both share electrons to satisfy
Answer: the valence electron for phosphorus is 5. To achieve an octet electron arrangement, it needs to lose 5 electrons or gain 3 electrons. It is easier to gain 3 electrons than to lose 5 electrons. So phosphorus has to gain 3 electrons. Explanation: Hope it helps sorry if it doesn't. In general, metals will lose electrons to become a positive cation and nonmetals will gain electrons to become a negative anion. Hydrogen is an exception, as it will usually lose its electron. Metalloids and some metals can be can lose or gain electrons. This is not always true, as elements such as nitrogen can lose electrons to become positive.
Br Lose Or Gain Electrons
You can view more similar questions or ask a new question.
